[qwiz qrecord_id=”aglimme-Unit4-4″]
[q]The first few questions involve the reaction between magnesium and hydrochloric acid. You may find it useful to write down the equation.
[q labels=”bottom”]Balance the following chemical equation:
___Mg(s)+___HCl(aq) ⟶___MgCl2(aq)+___H2(g)
[l]1
[fx] No. Please try again.
[f*] Great!
[l]2
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]3
[fx] No, that’s not correct. Please try again.
[f*] Great!
[l]4
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[q]If 4 moles of HCl react with excess Mg how many moles of H2 will be formed?
[c]1
[c*]2
[c]4
[c]8
[q labels=”bottom”]Fill in the correct ratios to set up the calculation to determine the moles of H2 produced by the reaction of 5.00g of Mg with excess HCl.

[l]5.00g Mg
[fx] No. Please try again.
[f*] Good!
[l]1 mol Mg
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]1 mol H2
[fx] No. Please try again.
[f*] Excellent!
[l]24.30g Mg
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[q labels=”bottom”]Fill in the correct ratios to set up the calculation to determine the grams of HCl required to produce 0.78g of H2.
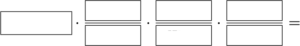
[l]1 mol HCl
[fx] No. Please try again.
[f*] Great!
[l]1 mol H2
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]2 mol HCl
[fx] No. Please try again.
[f*] Great!
[l]2.016g H2
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]36.46g HCl
[fx] No. Please try again.
[f*] Great!
[l]0.78g H2
[fx] No. Please try again.
[f*] Correct!
[/qwiz]