[qwiz qrecord_id=”aglimme-Unit2-4″]
[q labels=”bottom”]Place the correct variables for the equation that relates the energy of a photon to planks constant and the frequency.

[l]E
[fx] No. Please try again.
[f*] Correct!
[l]h
[fx] No, that’s not correct. Please try again.
[f*] Good!
[l]c
[fx] This equation does not use c.
[f*] Correct!
[l]λ
[fx] This equation does not use λ.
[f*] Great!
[l]ν
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[q labels=”bottom”]Place the correct variables for the equation that relates the energy of a photon to planks constant, the speed of light and the wavelength.
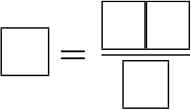
[l]E
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]h
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]c
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]λ
[fx] No, that’s not correct. Please try again.
[f*] Great!
[l]ν
[fx] This equation does not use ν.
[f*] Good!
[q]A bright violet line occurs at 435.8 nm in the emission spectrum of mercury vapor. What amount of energy, in joules, must be released by an electron in a mercury atom to produce a photon of this light?
[textentry minlength=”1″ use_dict=”false” use_terms=”false”]
[c]4.558•10-28J
[c]2.888•10-31J
[c]1.307•1011J
[c*]4.558•10-19J
[q]Heated lithium atoms emit photons of light with an energy of 2.961•10−19 J. Calculate the frequency of one of these photons.
[textentry minlength=”1″ use_dict=”false” use_terms=”false”]
[c*]4.469•1014 s-1
[c]1.491•106 s-1
[c]2.238•10-15 s-1
[c]1.962•10-52 s-1
[/qwiz]